Qimono the complete closed system for safe delivery of cytotoxic drugs
Stay safe with Qimono
It is widely documented that exposure to Systemic Anti-Cancer Therapy (SACT), can pose a significant health risk to patients during treatment as well as healthcare workers involved in the preparation and administration of the drugs .
Many SACT agents are known to be carcinogenic, teratogenic and mutagenic. Exposure may be through skin contact, skin absorption, inhalation of aerosols and drug particles as well as ingestion and needlestick injuries. This can occur at various points of the treatment process: during the preparation of the medication, whilst the drugs are administered and after therapy .
Adverse health effects reported by chemotherapy nurses include headaches, dizziness, nausea and hair loss.
Description of the device
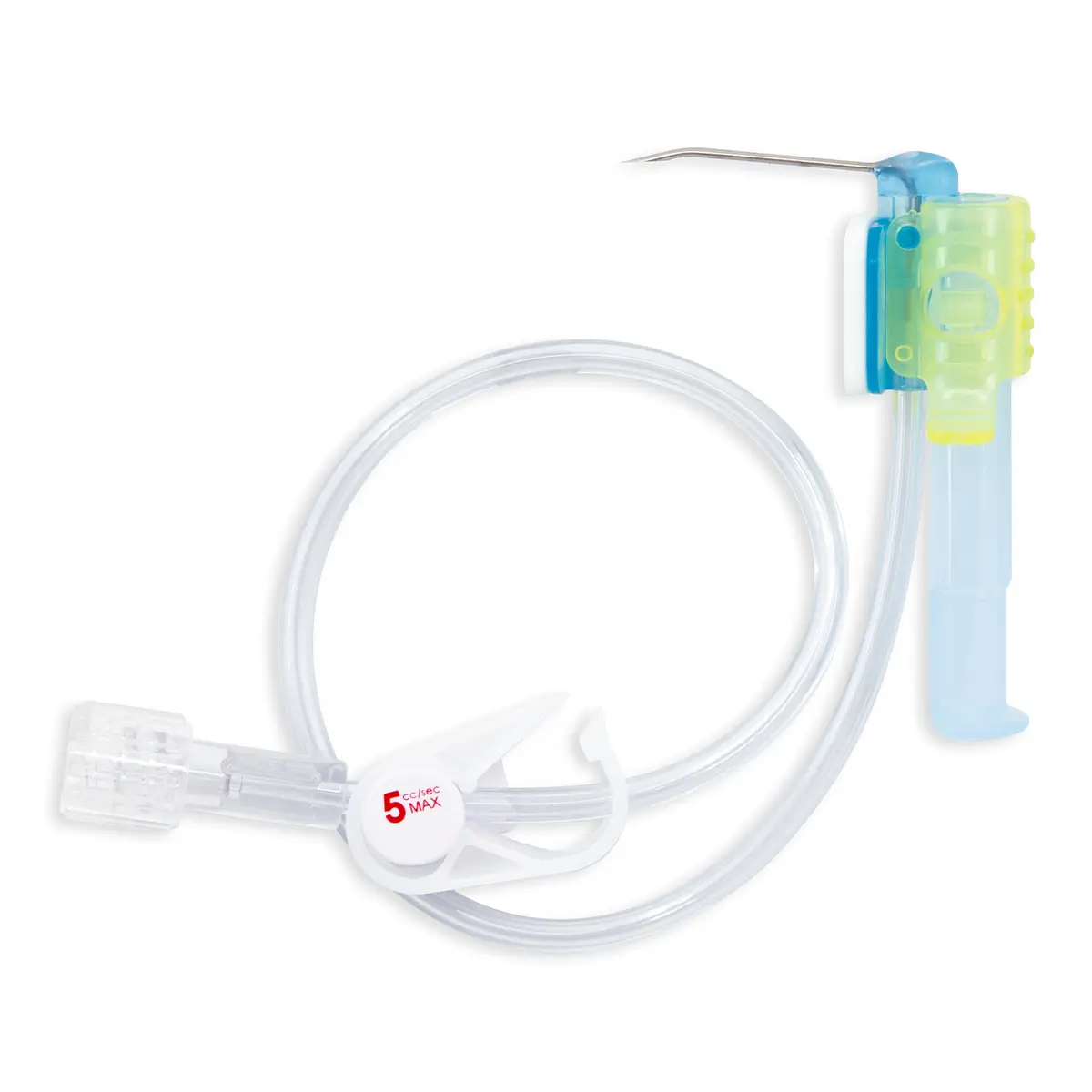
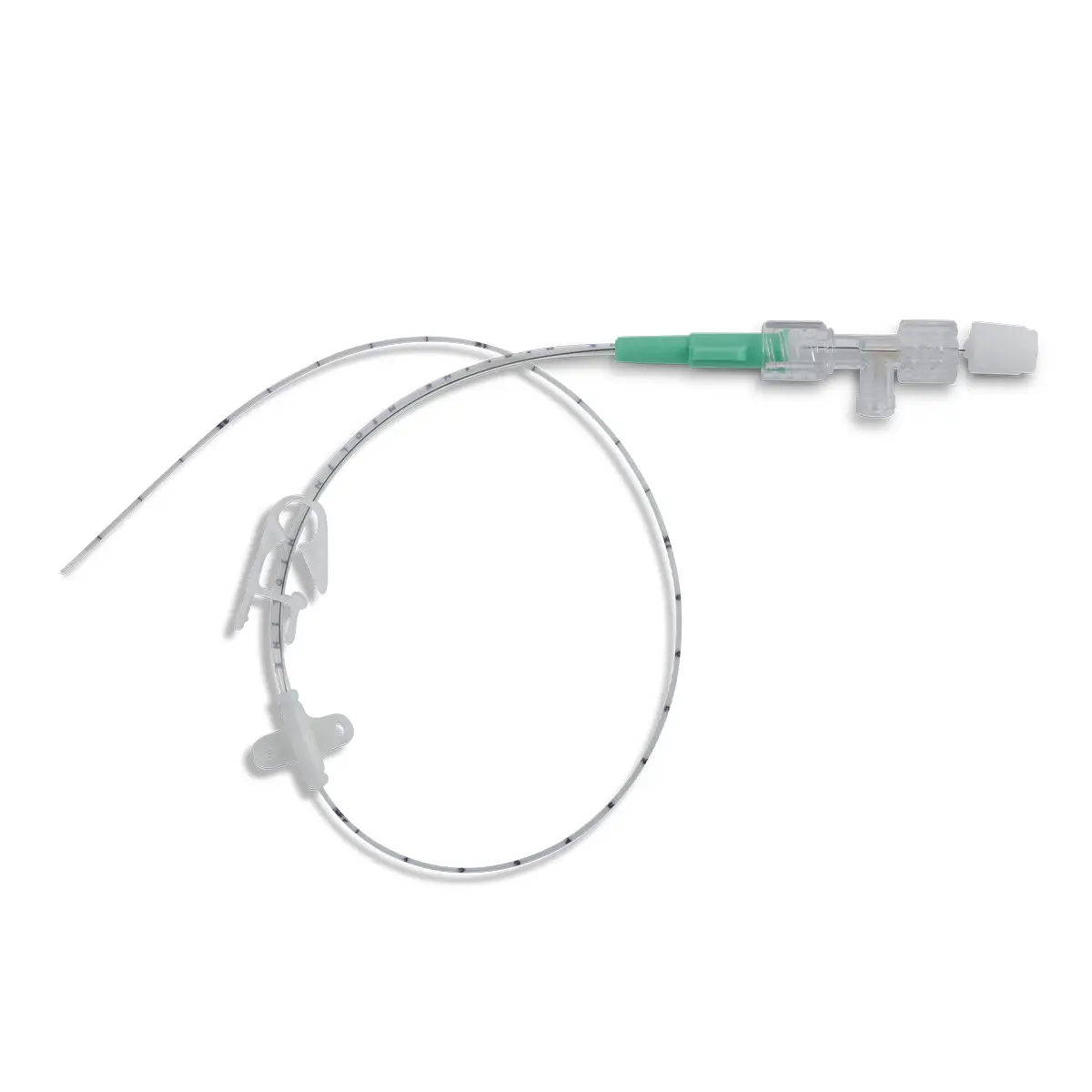
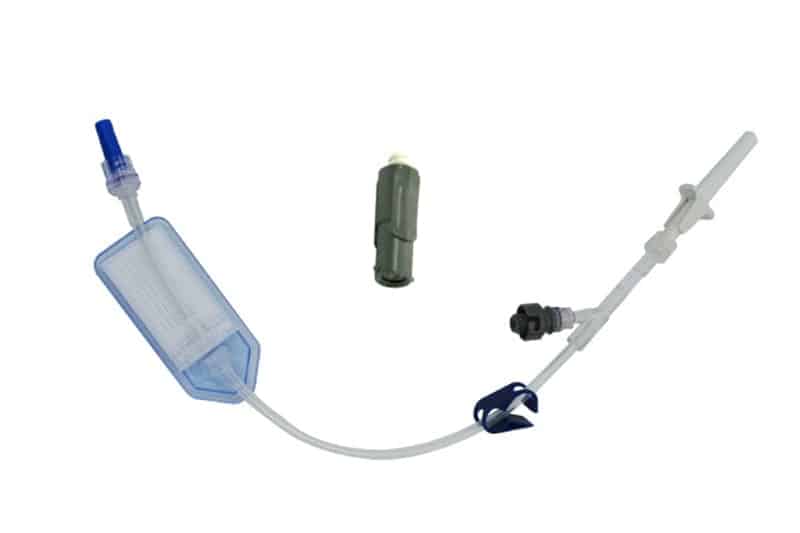
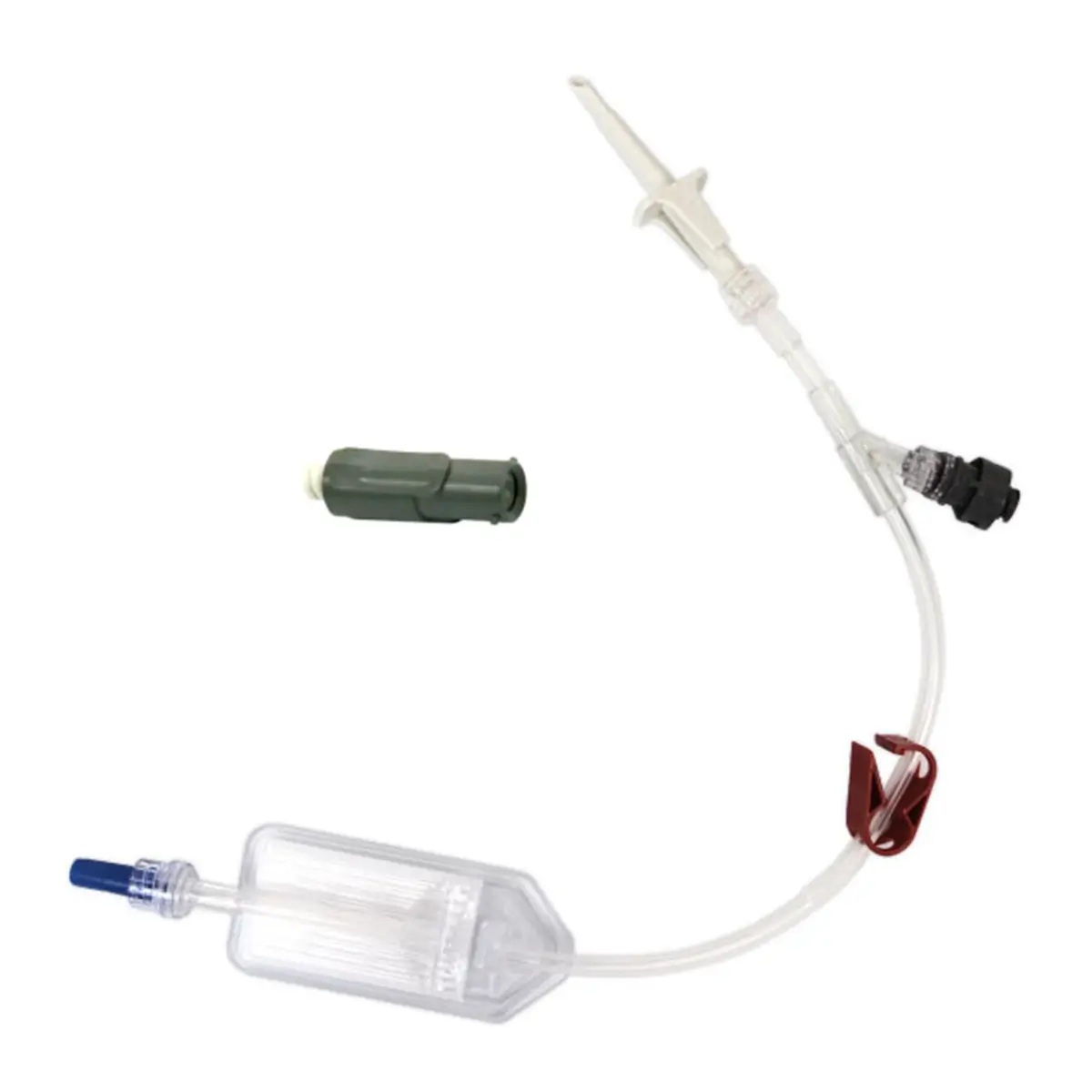
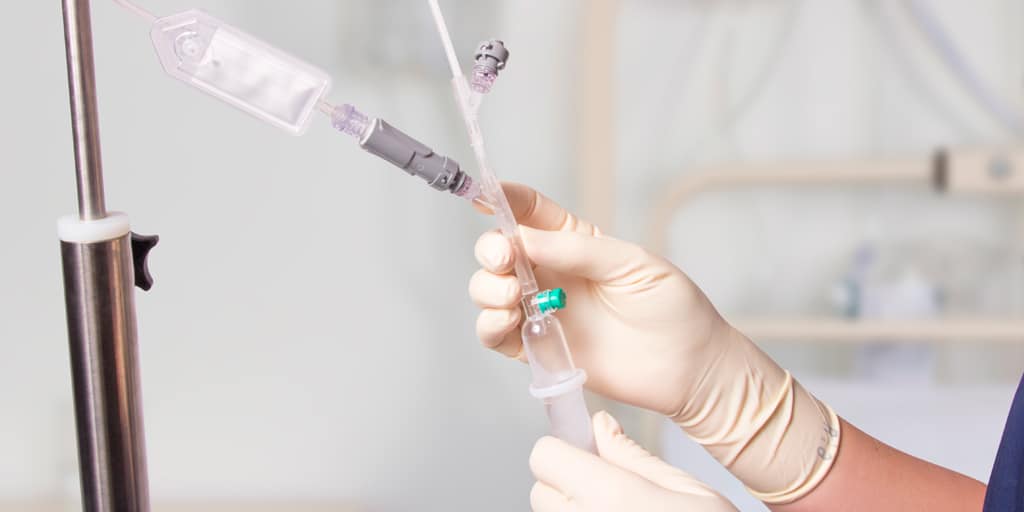
QimoConnect is an extension tube with Qimo Female and Qimo Male ports for pharmacy drugs dilution and administration, with or without filter.
QimoConnect includes the following components:
– 1 spike,
– 1 Qimo Female needleless connector,
– 1 extension tube with clamp,
– 1 Qimo Male needleless connector which must be attached to the distal end of the extension tube after priming of QimoConnect,
– with or without filter ( see hereunder).
PE-lined tubing with external layer in PVC, intermediate layer in EVA and internal layer in PE (featuring chemical inertia and good chemical inertness to medications).
Codes 7221.01 / 7221.020*: QimoConnect
Codes 7221.011 / 7221.0110*: QimoConnect filter (0.2 µm) – See the corresponding technical file
Codes 7221.012 / 7221.0120*: QimoConnect air filter – See the corresponding technical file
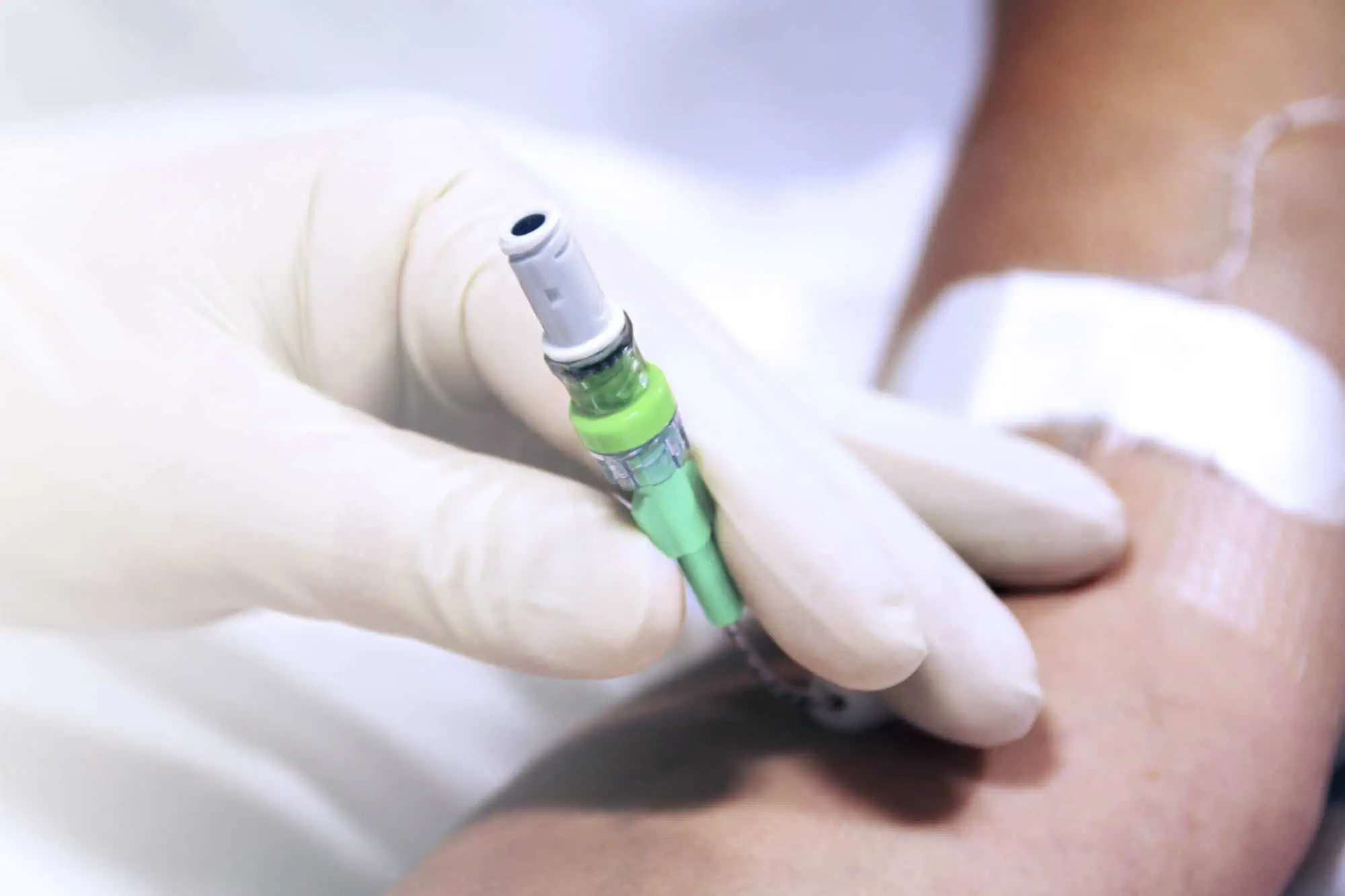
Qimo Female is a multi-purpose, closed, needle-free, connection device. See the corresponding technical file code 7210.02.
Qimo Male is a multi-purpose, closed, needle-free, I.V. connection device which has been designed for the preparation and safe administration of hazardous drugs. See the corresponding technical file code 7210.91.
Codes 7221.01 / 7221.011 / 7221.012: 1 unit in a supple blister.
Code 7221.020 / 7221.0110 / 7221.0120
– 7221.020: 10 units in a blister / 100 units in a box / 600 units in a case
– 7221.0110: 10 units in a blister / 100 units in a box / 600 units in a case
– 7221.0120: 10 units in a blister / 100 units in a box / 600 units in a case
* Codes 7221.020 / 7221.0110 / 7221.0120 are supplied without Qimo Male.