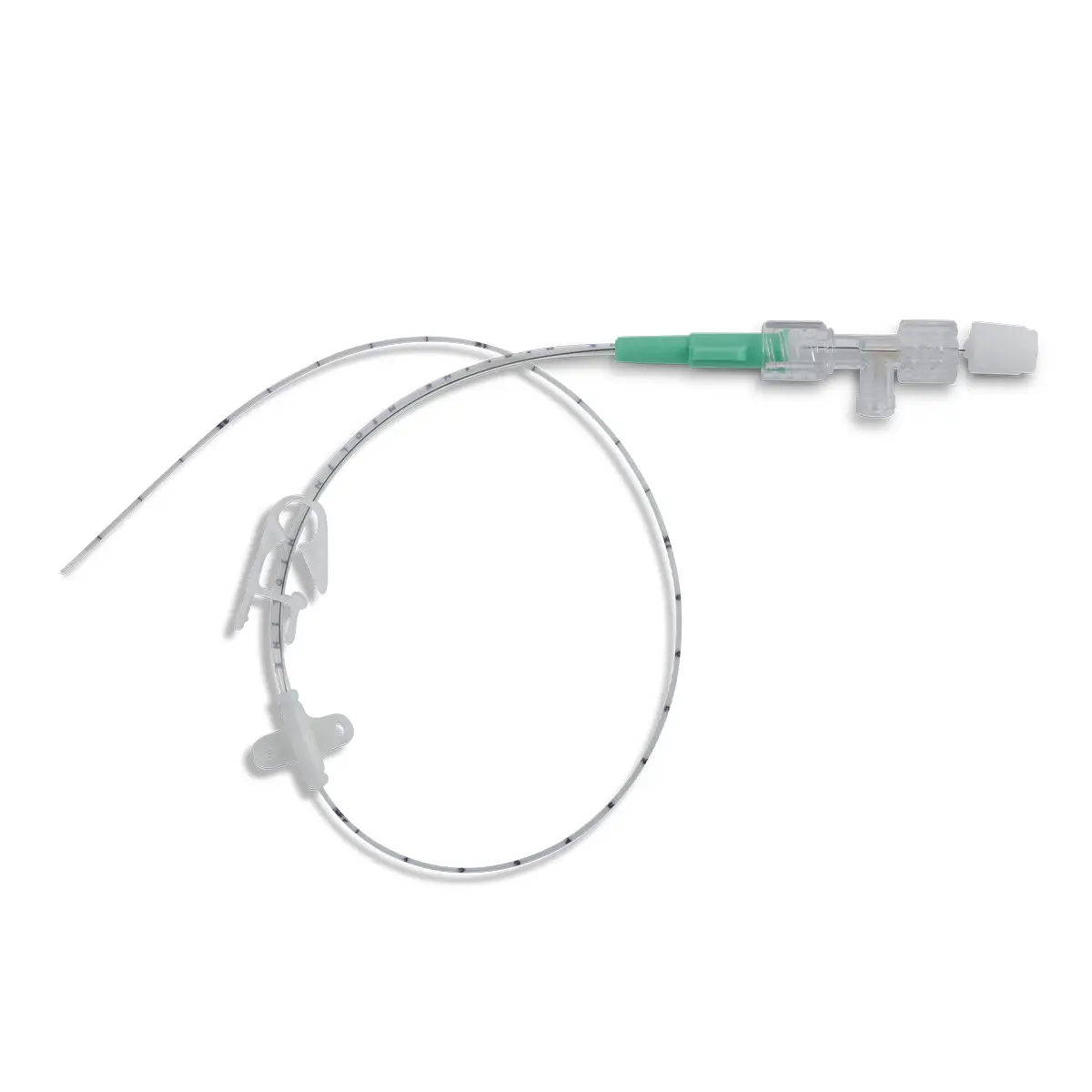
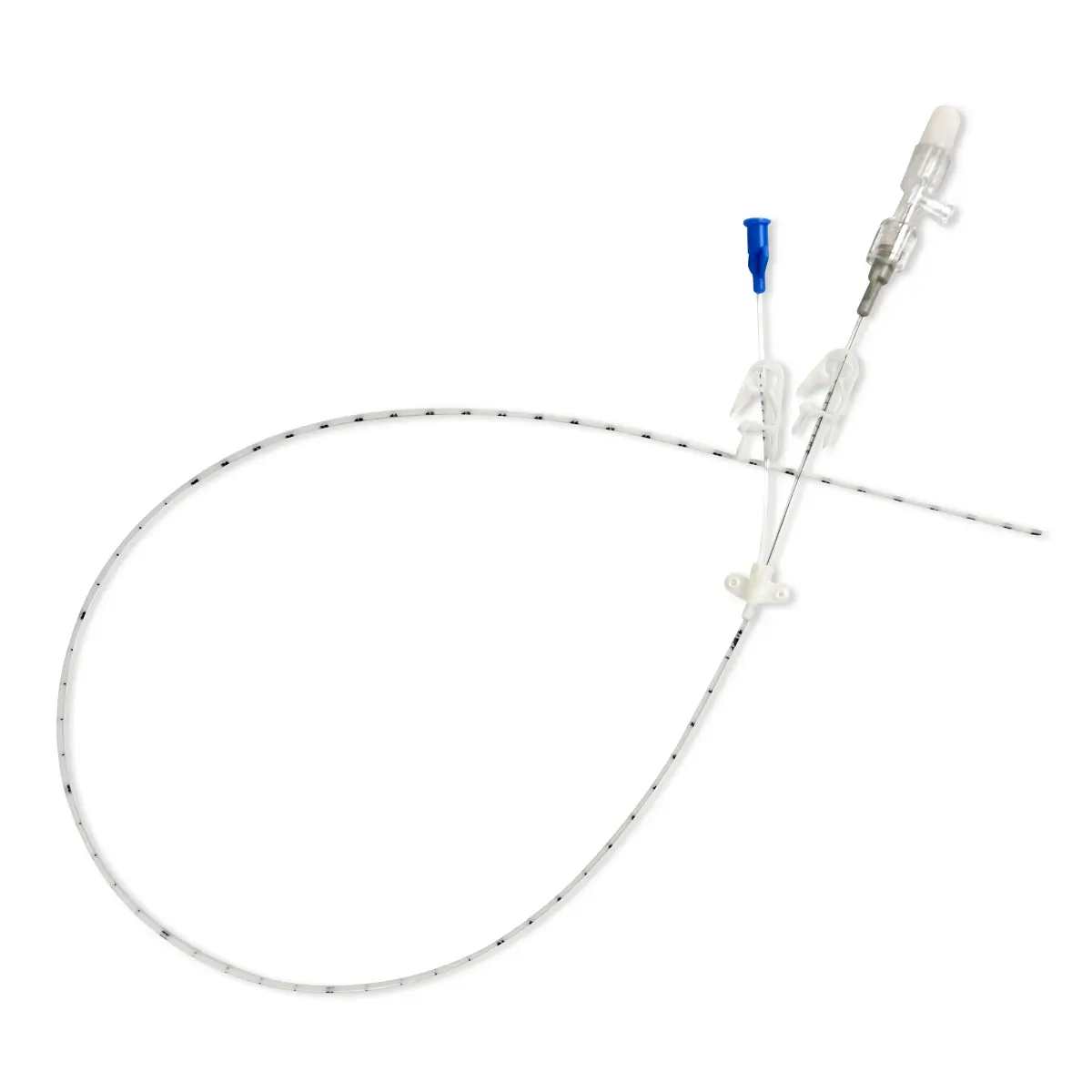
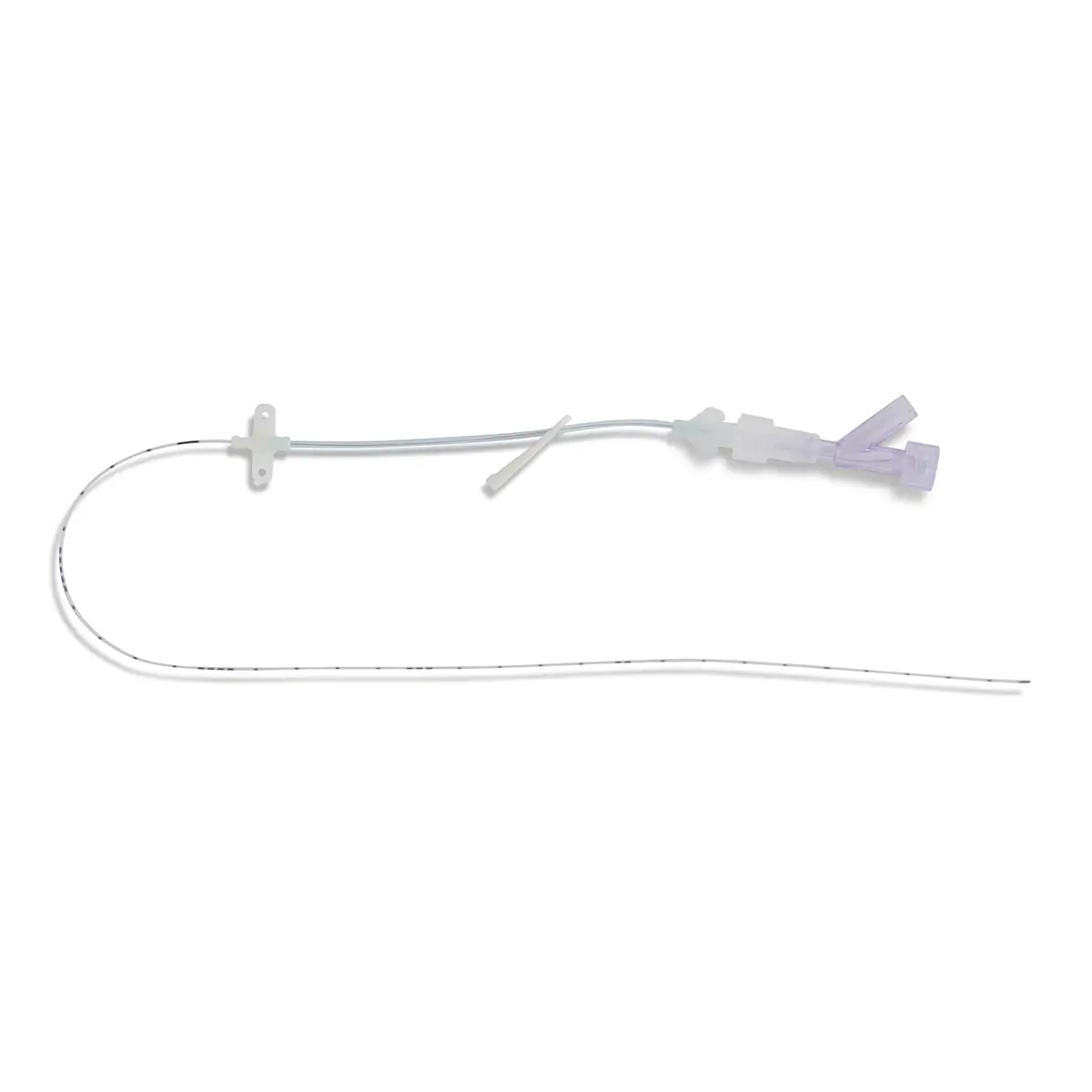
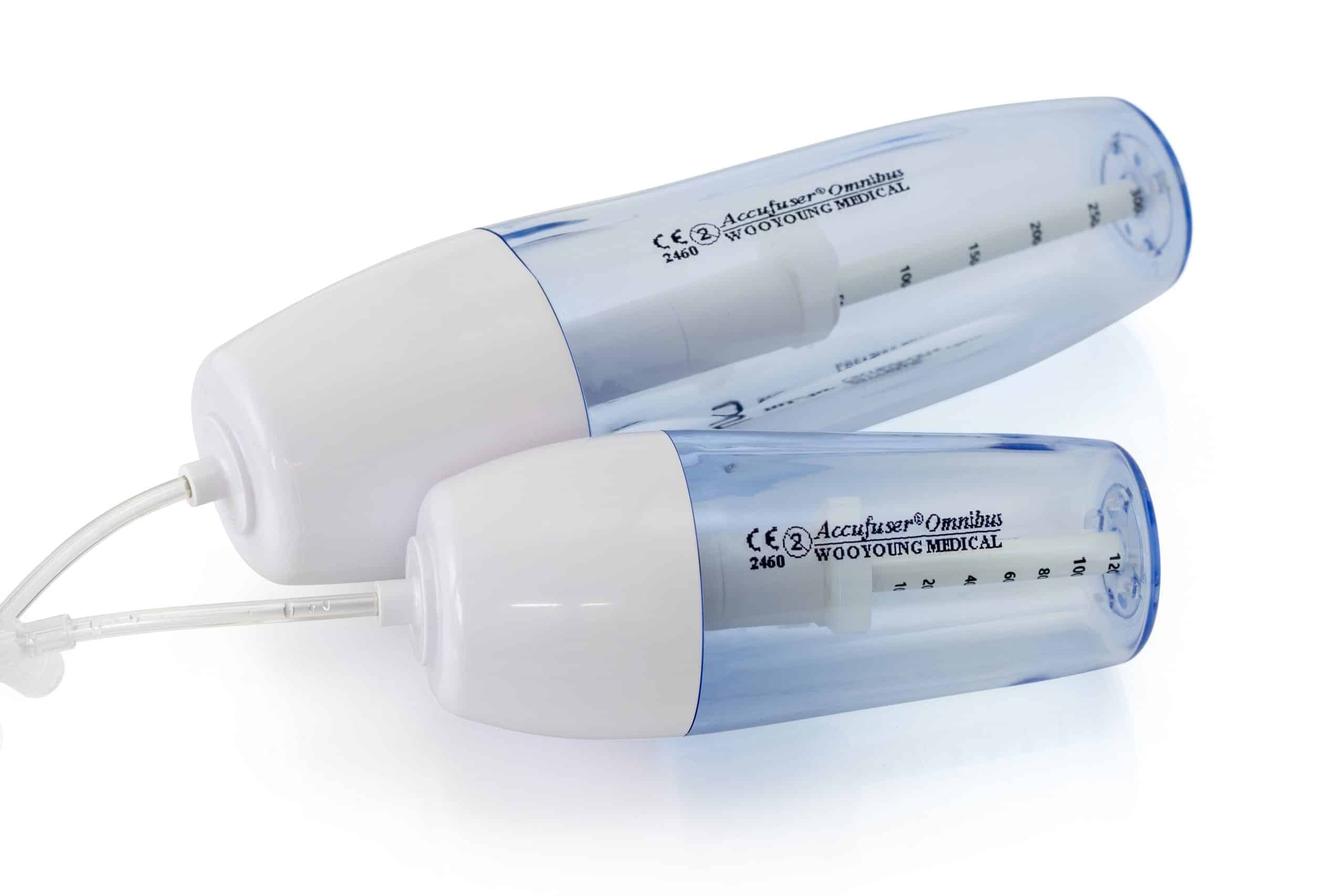
polysite™ is an easily administered implantable micro lightweight, hybrid port for paediatrics and brachial placement. It is positioned directly beneath the skin, usually in a discrete easily accessible place (chest level). Medicines then reach the blood circulation directly after simply puncturing the site through the skin.
- Hybrid: titanium and POM(1) port paediatrics and brachial placement
- Profile-shaped design
- Easy to connect
- Titanium-POM combination: low weight
- Titanium reservoir: compatibility with antineoplastic agents
- Radiopaque connecting ring
- MRI conditional & CT compatible