Vygon Freedom 5 is a mechanical single patient device for patient controlled analgesia. It can be used in the Accident and Emergency department, for post-operative pain in the recovery room or for pain treatment in sickle cell disease.
If necessary, Freedom 5 can be refilled twice. In this case a refill kit is available.
The simplified refill kit code 7302.01 (for PCA code 7301.10 or 7301.12) and 7302.02 (for PCA code 7301.05, 7301.50 ou 7301.501) contains:
– 1 PCA lockable capping device with lever and cord
– 1 female/female Luer-lock adaptor
A complete refill kit with 60ml calilbrated reservoir is available under code 7301.60 and 7301.62: see corresponding technical file.
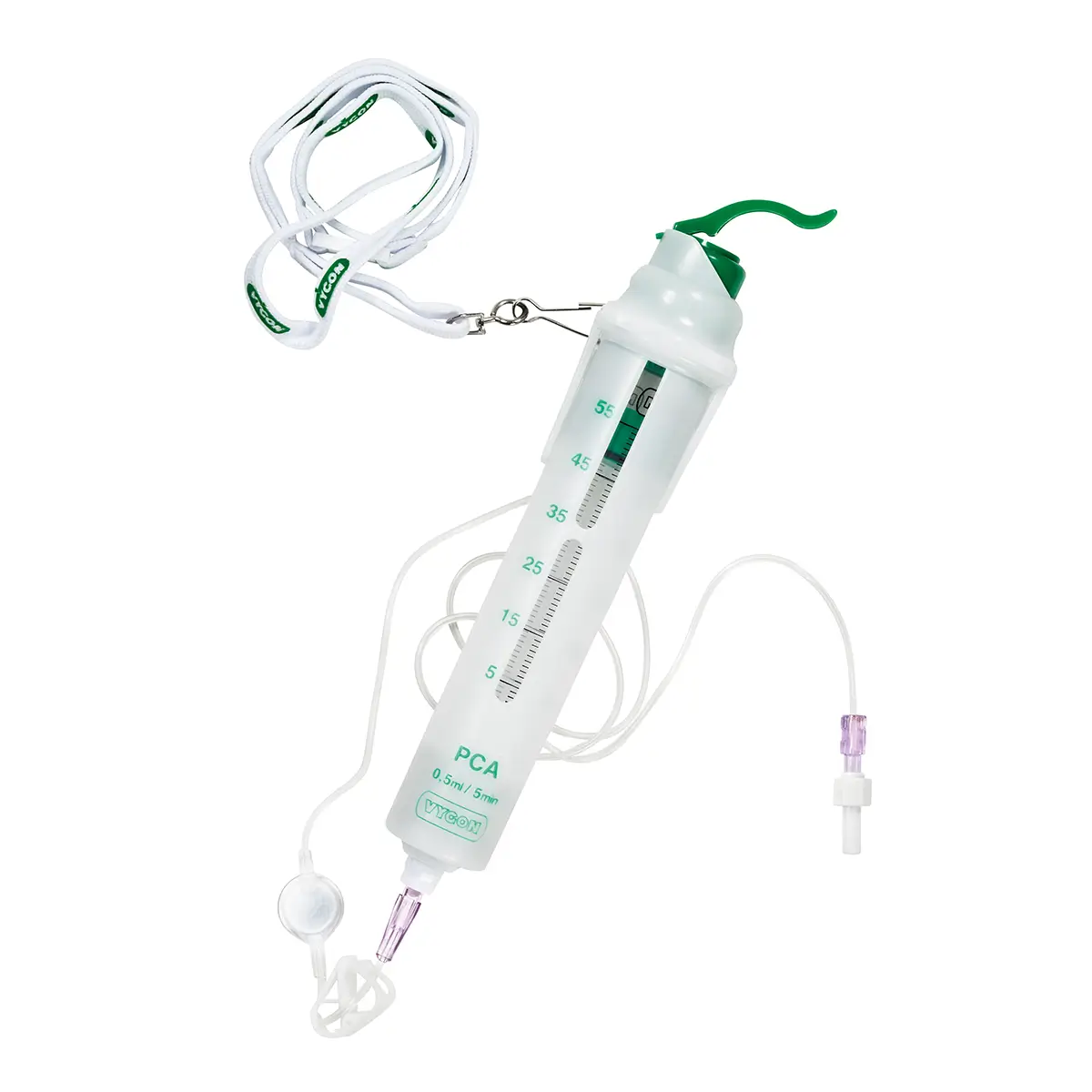
The Freedom 5 kit contains:
– 1 PCA reservoir carrier with inspection window to check the amount of morphine remaining in the reservoir. A bolus chamber of 1 ml or 0.5 ml (depending on the model) is located at the bottom part of the reservoir carrier
– 1 calibrated reservoir (55 ml). This reservoir must be locked within the carrier making sure that the graduations of the reservoir are visible in the inspection window of the carrier
– 1 capping device with lever
– 1 attachment cord
– 1 polyethylene storage bag
– 1 female/female Luer-lock adaptor (code 892) for filling the reservoir with a syringe
– 1 female Luer-lock obturator (code 888) for temporary capping of a filled reservoir
– 1 label for the PCA reservoir carrier
– 1 single-lumen extension tube (length 125 cm) with antibacterial and air-eliminating filter and anti-siphon valve: model 7301.10
or
– 1 “Y” extension tube (length 90 cm) with antibacterial and air-eliminating filter and anti-siphon valve (lumen 1) / non-return valve (lumen 2): model 7301.12